Types of Filter Integrity Testing

Filter integrity testing is performed to assure a contamination-free product. In this, the integrity of the filter membrane, disk filters, capsules/cartridges are checked.
The machine that performs this task is called filter integrity machine. It needs to comply with Class B and Digital Devices under Part 15 of FCC regulations. These machines generally work on the principle of differential pressure.
This test equipment specifically has a in-built data description management unit that enables data management and report generation.
The performance of any sterilizing filter is directly associated with the retention of bacterias below 0.22 µm. So, different size filters are used in sequence to reduce the load on a single small-sized filter.
The first filter has a 0.45 µm size followed by 0.22 µm. Later, they get sterilized with steam for another batch process. These filters require filter integrity testing both before and after use if tested non-destructively.
The two cases to perform filter integrity testing are:
- New Filters
- Filters that require re-use
In the first case, filter manufacturers themselves perform the testing along with certificates and documentation. If so, it is not mandatory for drugmakers to test new filters. However, they may do to have more confidence.
Meanwhile, the second case often requires testing to check the intactness of the pore size of the membrane. Testing the differential pressure helps to assure the integrity of the filter. Agar plates re-check that no bacteria crossed the 0.22 µm filter.
Table of Content
Classification of Filter Integrity Testing
The FDA set forth guidelines and requirements to conduct and document filter integrity testing.
The testing is classified into two categories:
- Destructive Testing
- Non-Destructive Testing
Destructive Testing
Destructive Testing is by and large carried out at the filter manufacturer’s location from each lot as per their pre-defined sampling plan.
Destruction of filters takes place to ensure that no bacteria can pass through the membrane. Simply put, filters retain bacteria.
This testing complies according to the method outlined in ASTM F838-05 (formerly ASTM F838-83), Standard Test Method for Determining Bacterial Retention of Membrane Filters Utilized for Liquid Filtration.
Both Health Industry Manufacturer’s Association (HIMA) and FDA have defined the retention of the challenge bacteria level to be 107 per cm2.
The FDA considers a filter as “Sterile” when it retains the challenge of bacteria Brevundimonas Dimunita (ATCC 19146) at least 1×107 per cm2 of filtration area. For more information, see an insight into membrane filter validation.
Non-Destructive Testing
Filters used in the previous batch may be damaged or distorted ultimately posing questions on their integrity for the past and coming batch.
Reusing a filter adopts non-destructive testing where it is checked for its intactness and quality for the next batch.
If filter integrity testing fails, the previous batch may need re-testing/processing as per suitable immediate corrective action. The filter also becomes useless and hence discarded.
The following are the different methods for non-destructive filter integrity testing:
- Bubble Point Test
- Diffusion Test
- Pressure Drop Test
- Water Intrusion Test
These tests were developed to physically make sure the integrity of a filter cartridge. Each test has its own significance and working mechanism explained below.
Bubble Point Test
This test works on the principle of capillary action and surface tension.
Liquid when passed through the filter, fills inside the pores with the help of surface tension and capillary action.
The minimum amount of pressure required to remove the liquid out from the pores helps measure the pore size. The pressure is applied accordingly.
Nowadays testing units have pre-loaded automated sequences for each test.
The bubble point test goes like this:
- Filter is wetted with suitable solvent (‘water for hydrophilic filters’ and ‘water+alcohol for hydrophobic filters’) uniformly.
- Pressure is applied to the upstream of the filter. The amount of minimum pressure expected from the recommendations provided by filter manufacturers.
- Pressure increases slowly until bubbling occurs downstream.
If bubbling appears at a lower pressure than the set pressure, the test is considered a fail. The pressure at which bubbling occurs downstream of the filter is its Bubble Point.
Possible reasons for failure:
- Incorrect installation of the filter inside the housing
- Filter wetting is non-uniform
- The unsuitable solvent used for wetting the filter is causing compatibility issues due to different surface tension than recommended or;
- Filter failed integrity due to deformation
Once we get the bubbling point, the diameter of the pore is calculated by:
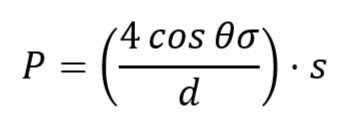
where,
P – min. pressure when bubbling occurred
d – Pore dia.
θ – Contact angle between solid and liquid (this angle formed because of surface tension and capillarity)
s – correction factor
σ – surface tension
Don’t forget to maintain unit consistency.
Diffusion Test
This test works on the principle of Fick’s Law of Diffusion, where the rate of diffusion is directly proportional to the membrane’s surface area.
The effectiveness of the diffusion test depends on the downstream gas flow rate, unlike the bubble point test where pressure was the key aspect.
Typically, the pressure used to perform this test is 75-80% of the bubble point of that membrane. The manufacturer provides requirements/specifications for this test.
The procedure goes like this:
- Filter is wetted uniformly with a suitable solvent.
- Pressure is applied upstream of the membrane, gradually increased up to the recommended value and allowed for stabilization.
- Downstream gas flowrate is measured with the help of a suitable flow meter for 60 seconds.
If the value of the gas flowrate comes higher than the recommendation, the test failed.
Possible reasons for failure:
- Incomplete wetted membrane
- Insufficient stabilization
- Filter failed integrity due to damaged pore size
Verification of the test is done by using,
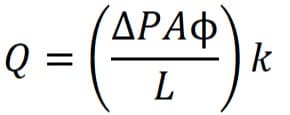
where;
Q – Diffusional flow
A – Membrane surface area
L – Effective Length
ΔP – Differential Pressure
Φ – Porosity of the membrane
k – Diffusivity
Again, don’t forget to maintain unit consistency. This test is recommended when handling high-surface-area systems starting from 1.5 m2 onwards.
Pressure Drop Test
While dealing with different housing arrangements, bubble point or diffusion tests becomes ineffective.
Then, pressure drop tests are performed on the automated filter integrity testing units.
Because this test is performed upstream of the assembly, the downstream manual operations get eliminated.
As manufacturers do not specify the exact test requirements, this test relies on system volume data. They measure changes in upstream pressure because of diffusion via the application of an appropriate and accurate pressure gauge.
Maximum allowed pressure drop:
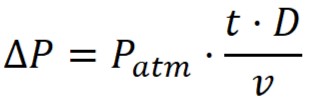
where;
D – Rate of Diffusion
Patm – Atmospheric Pressure
V – Volume of system upstream
t – time
ΔP – Pressure Drop or Decay
Water Intrusion Test
This test is typically performed to detect the quality of defects of hydrophobic filters. These filters are installed as air vent filters.
Here’s the article that explains water intrusion test requirements.
Typical Arrangement
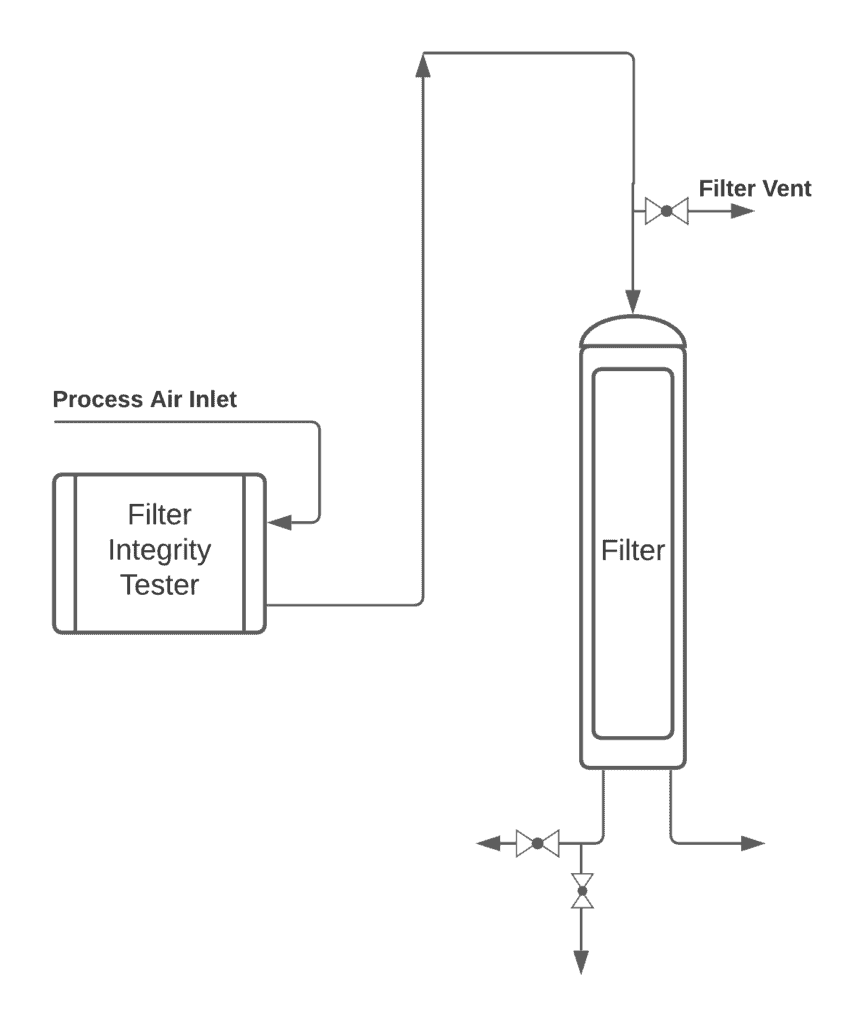
Generally, common components installed on the testing unit are housing, HMI, printer, tubings, alarms, TC Clamp connections, etc.
Proper connections at the inlet and outlet prior to starting the test sequence must be checked before initiating.
Check the proper recipe is set, loaded, and ready to execute.
Automation Aspects for Filter Integrity Units
The reliability of the testing unit components depends on their MoC. Hence, compatibility with the laboratories and commercial manufacturing environments is important.
The manufacturer takes care of that part, including automation hardware.
To prevent mishandling during emergency conditions, alarms and interlocks should be considered.
The system prevents any unauthorized access to recipe modification, start and stop sequence, and other functions.
An audit trail is another requirement that captures all the activities any user performs. Secure backup and restore procedures are also included in the automation package.
GxP Requirement for Filter Integrity Units
- 21 CFR Part 11, Electronic Records, and Signatures
- GAMP 5 A Risk-based Approach to compliant GxP Computerized Systems
- BS EN 60204-1, Safety Guidance and Recommendations on Electrical Equipment
- Certificate of Compliance
Some test units produce paper-based records storing no data electronically. Such units don’t fall under the requirements of 21 CFR Part 11. Also, the application of such units is not designed for cGMP-regulated environments.
For testing units requiring compliance with FDA 21 CFR Part 11, the validation plan should cover procedural and technological control.
Consider the following points while complying with 21 CFR Part 11:
- Generation, Modification, and Maintenance of Electronic Records
- Audit Trails for all the Electronic Records
- Equipment Qualification and Test Method Validation
- Training of Concerned Personnel for 21 CFR Part 11
- Electronic Signatures
- User Management
- Data Export in the format suggested by FDA
- Human Readability for defined retention time
Filter Integrity Test: FAQs
What is filter integrity test?
Tests carried out to verify and assure the quality and readiness of the filter membrane regarding the regulatory requirements are called filter integrity tests. If failed, shows that the filter is no longer re-usable, and also the previously filtered batch needs inspection.
How does a filter integrity tester work?
As mentioned previously in this article, two basic classifications include Destructive and Non-Destructive testing. Non-Destructive tests include testing based on Bubble Point, Diffusion, and Pressure Drop tests as suitable to the requirements.
What is bubble point test for filters?
This test works on the principle of capillary action and surface tension. Liquid when passed through the filter pores fills inside the pores with the help of surface tension and capillary action. Pressure is applied to measure the diameter of a pore. The minimum amount of pressure required to remove the liquid from the pores helps in measuring the pore size.
What is water intrusion test?
This test is typically performed to detect the quality of defects in hydrophobic filters. These filters are generally installed as air vent filters. Water with pressure penetrated inside the filter upstream to compare the outcomes with the manufacturer’s specification limits.
What does bubble point mean?
The minimum amount of pressure at which the first bubble of liquid comes out from a pore of the membrane, known as the bubble point for that filter.
What does a 0.22 micron filter remove?
The diameter or size of the pore is uniformly maintained on the filter membrane. We consider this size one of the smallest in the industry to eliminate the finest-sized particles. This is effective in removing bacteria and not viruses.

Dear Saket,
If I just have information of Flow rate and pressure drop, no integrity testing information. how I can calculate and set up integrity testing for this filter. I have a machine for integrity testing (IT5 of Merck)
“Flow rate and pressure drop:
sample met maximum pressure drop of 6 psi (410mbar) at 10 gmp ( 38L / min) per 10in. equivalent with water at 23oC”
Thank you so much!
Great article. Auditors are expecting filter integrity validations and sending used filters for filter integrity testing.
Is it just to use the smallest bacteria at 0.22 micron and it’s able to filter it?
Thanks
Absolutely, Hale! And yes, you need to validate that.
Nice way of explanation
Glad to know that, Akshay!
Great efforts Saket!!!
Loved your articles
Keep up the good work 👍🏽
Hey Sagar! Really appreciate your words 🙂 Stay tuned for more.
Helpful knowledge. Works smoothly as per interview aspect.
Hey Vishal! Glad you find this helpful. Keep in touch and stay tuned.
Hi.. Helpful article for overall understanding. Can yo share the calculator for these tests as you shared F0 value calculator. That would be of great help. Thank you.
Hi Rishu, Glad that this article helped you. Definitely yes, I am working on some other aspects right now. So I’ll do this too in coming time. Till then, stay tuned.